The slight difference in atomic weight between the uranium-235 and uranium-238 isotopes figured greatly in nuclear physics during the 1930s and 1940s. 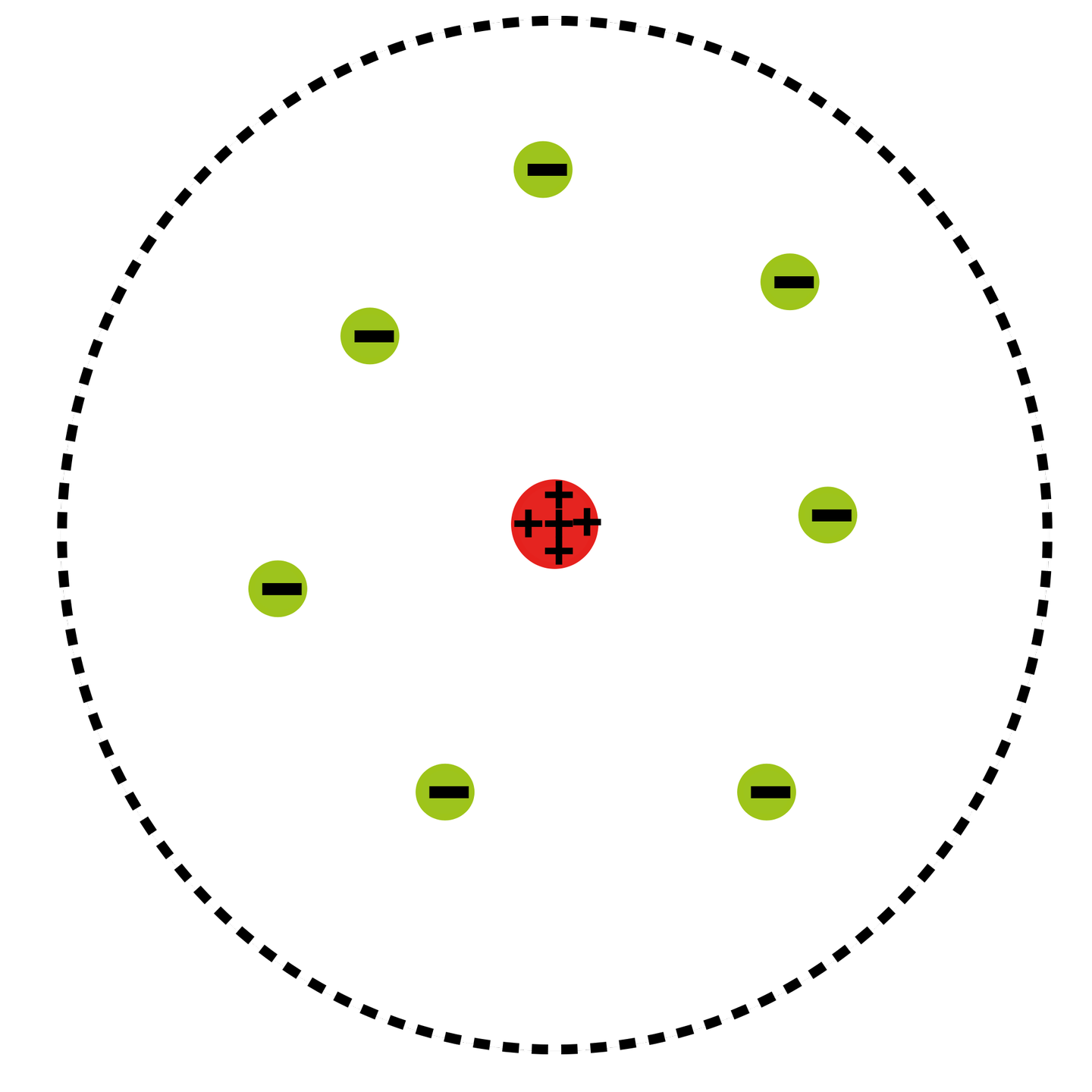
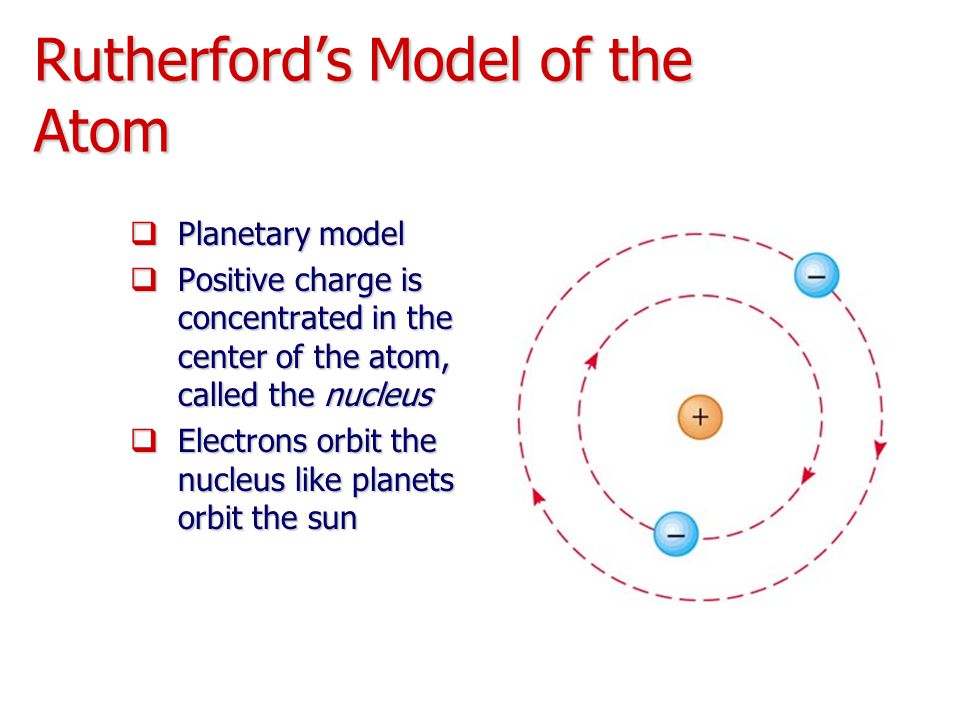
But uranium-238, which accounts for over ninety-nine percent of natural uranium, has 146 neutrons in its nucleus, compared with 143 neutrons in the rare uranium-235 (.7 percent of natural uranium) and 142 neutrons in uranium-234, which is found only in traces in the heavy metal. The three isotopes of uranium, for instance, all have ninety-two protons in their nuclei and ninety-two electrons in orbit. These different classes of atoms of the same element but with varying numbers of neutrons were designated isotopes. Scientists found that the weight discrepancy between atoms of the same element resulted because they contained different numbers of neutrons. It was Chadwick's discovery of the neutron in 1932 that explained this mystery. This simple scheme became more complicated when chemists discovered that many elements existed at different weights even while displaying identical chemical properties. Hydrogen, with one proton, came first and uranium, with ninety-two protons, last on the periodic table. The number of protons determined the element's atomic number. Another addition came in 1932 when James Chadwick, Rutherford's colleague at Cambridge, identified a third particle, the neutron, so-named because it had no charge.īy the early 1930s the atom was thought to consist of a positively charged nucleus, containing both protons and neutrons, circled by negatively charged electrons equal in number to the protons in the nucleus. The proton, as this subatomic particle was named, joined the electron in the miniature solar system. In the process of changing nitrogen into oxygen, Rutherford detected a high-energy particle with a positive charge that proved to be a hydrogen nucleus. At the time of Rutherford's breakthrough, the atom was conceived as a miniature solar system, with extremely light negatively charged particles, called electrons, in orbit around the much heavier positively charged nucleus. The road to the atomic bomb began in 1919 when the New Zealander Ernest Rutherford, working in the Cavendish Laboratory at Cambridge University in England, achieved the first artificial transmutation of an element when he changed several atoms of nitrogen into oxygen. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
